The chemistry and physics of bio-sensory systems.
Normally I want to see the entire course syllabus laid out before me, but I am glad we cannot see too far in advance. The reason for my feeling this way is that sometimes we teachers assume we know everything about something and might not treat the material as seriously as we should. This week totally fascinated me, first because I can SEE HOW EVERYTHING WE LEARNED SINCE MODULE 1 connects, flows and builds upon each other. See my individual glossary term from this week, micro cantilevers, for a full description.
One article from this week's reading that really helped tie in biological phenomena with sensory systems is the Wasabli-protein receptors in the snake that sense heat. Here is a link to the article:
One article from this week's reading that really helped tie in biological phenomena with sensory systems is the Wasabli-protein receptors in the snake that sense heat. Here is a link to the article:
Another awesome link is to an article that discusses how the sea urchin uses its entire body as an eye. I found this particularly interesting because we do an embryology/developmental biology unit in my AP Biology class and we use live sea urchins. This link provides some very useful extension information that I plan on using with my own students:
Introduction:
In Module 3, I designed a nanorobot that initially resembled a cross between an octopus “sentinel”
(inspired from a real octopus and the machine form the movie “The Matrix”) and a bacterium.
The final design looked more like a bacterium with artificial flagella and rotary motors.
See Link to my original project here: The Sentinel Bio-inspired Medical Nanobot.
Medical nanotechnology is an emerging field inspired by the work of Richard Feynman. Dr.
Feynman recognized that living systems obey the Laws of chemistry and physics and encouraged
his graduate students model the efficiency of energy flow in living systems in the design of
machines. (Feynman: Tiny Machines You Tube Video). Nanobots have the potential to
revolutionize medicine and produce less invasive procedures for treatment and diagnosis of
many diseases, especially cancer. Miniaturization of functional devices down to nanoscale
dimensions has been enabled through the advancement and new development of both bottom up
and top down approaches in nanotechnology. (Schurle, S. 2012) My nanorobot was designed
for propulsion through the aqueous environment of the human bloodstream and is constructed of
either carbon nanofibers or modified nucleic acids so the machine would evade the immune
response. I have decided to stick with the medical nanorobot idea and add on to my original
design. The three sensor modalities that I have chosen include magnetoreceptors, thermosensors
and chemoreception. Each modality is discussed in a separate section below and includes the
benefits, limitations and bioinspiration for the each selection.
In Module 3, I designed a nanorobot that initially resembled a cross between an octopus “sentinel”
(inspired from a real octopus and the machine form the movie “The Matrix”) and a bacterium.
The final design looked more like a bacterium with artificial flagella and rotary motors.
See Link to my original project here: The Sentinel Bio-inspired Medical Nanobot.
Medical nanotechnology is an emerging field inspired by the work of Richard Feynman. Dr.
Feynman recognized that living systems obey the Laws of chemistry and physics and encouraged
his graduate students model the efficiency of energy flow in living systems in the design of
machines. (Feynman: Tiny Machines You Tube Video). Nanobots have the potential to
revolutionize medicine and produce less invasive procedures for treatment and diagnosis of
many diseases, especially cancer. Miniaturization of functional devices down to nanoscale
dimensions has been enabled through the advancement and new development of both bottom up
and top down approaches in nanotechnology. (Schurle, S. 2012) My nanorobot was designed
for propulsion through the aqueous environment of the human bloodstream and is constructed of
either carbon nanofibers or modified nucleic acids so the machine would evade the immune
response. I have decided to stick with the medical nanorobot idea and add on to my original
design. The three sensor modalities that I have chosen include magnetoreceptors, thermosensors
and chemoreception. Each modality is discussed in a separate section below and includes the
benefits, limitations and bioinspiration for the each selection.
1. Magnetoreception:
• Bio-inspiration: the articles from Module 4 reading provided the basis for this choice
along with some previous articles from my first robot design. I am fascinated with the use
of Earth’s magnetic field by animals to guide their taxis. The main sources include:
• Magnetoreceptor cells in vertebrates
• Turtles Use Earth’s Magnetic Field for migration
• Magnetotaxis in Honey Bees and Bacteria
Honeybees, turtles, bacteria have all been shown to navigate using a magnetic field as a
guide. (Mayroides, 2013). Investigations on the organisms mentioned above reveal the
presence of crystalline iron compounds that produce specific wave resonance in the
presence of external magnetic fields. This resonance in response to external magnetic
fields induces an electrical current that triggers either a “nerve” impulse or some sort of
electrical coupling event. (Kuterbach, 1986).
This short video clip shows a tiny nanobot responding to a magnetic field and was
published at the Institute of Robotics and Intelligent Systems at ETH Zurich
• Bio-inspiration: the articles from Module 4 reading provided the basis for this choice
along with some previous articles from my first robot design. I am fascinated with the use
of Earth’s magnetic field by animals to guide their taxis. The main sources include:
• Magnetoreceptor cells in vertebrates
• Turtles Use Earth’s Magnetic Field for migration
• Magnetotaxis in Honey Bees and Bacteria
Honeybees, turtles, bacteria have all been shown to navigate using a magnetic field as a
guide. (Mayroides, 2013). Investigations on the organisms mentioned above reveal the
presence of crystalline iron compounds that produce specific wave resonance in the
presence of external magnetic fields. This resonance in response to external magnetic
fields induces an electrical current that triggers either a “nerve” impulse or some sort of
electrical coupling event. (Kuterbach, 1986).
This short video clip shows a tiny nanobot responding to a magnetic field and was
published at the Institute of Robotics and Intelligent Systems at ETH Zurich
My nanobot will contain either a magnetic ferrofluid (as in my first post) or small
nanoplates composed of crystalline iron similar to that found in the magnetoreceptor cells
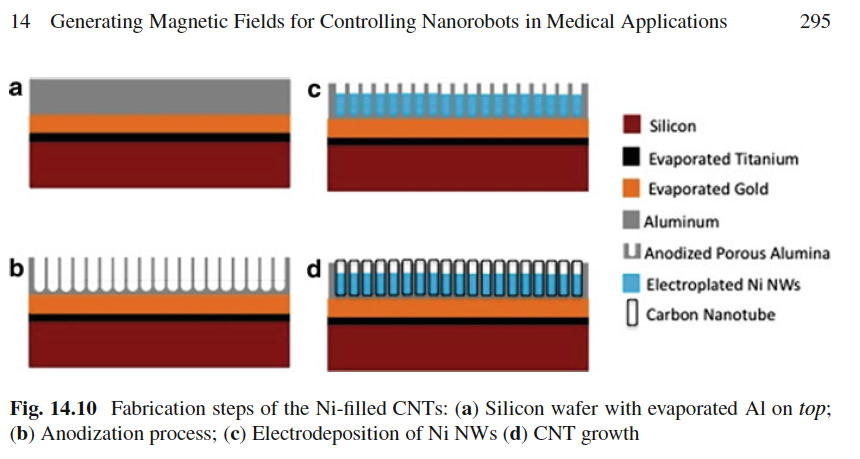
of turtles and honeybees. The crystals are applied to silicon plates. See a picture of the
process of making nanomagnetic particles similar to the crystals found in the
magnetoreceptor cells of insects and vertebrates:
nanoplates composed of crystalline iron similar to that found in the magnetoreceptor cells
of turtles and honeybees. The crystals are applied to silicon plates. See a picture of the
process of making nanomagnetic particles similar to the crystals found in the
magnetoreceptor cells of insects and vertebrates:
(Image: S. Schuerle, Nanorobotics, 2013).
Here is another image from Shurle showing the magnetic materials:
Here is another image from Shurle showing the magnetic materials:
• How will it work?
• The magnetic crystals will have two functions:
• 1. The crystals can be used to guide the device through the human body via an
applied magnetic field OR..
• 2. The crystals will detect and be attracted to a magnetic structure already within
the body.
Regarding Option #2, research has been done on producing biosensors that respond to
magnetic targets created by antibody ligand interaction. Cancer cells possess unique
surface receptors. Magnetically labeled antibodies (ferro) have been produced that attach
to cells, groups of cells in tumors creating strong magnetic fields that can be sensed by
the nanobot. The nanobot then can recognize very specific targets for some purpose such
as medicine delivery, or attach to the cells where an external field can then activate the
robot to heat up, release some cargo, ect. (See article Smith, 2011).
• Limitations:
• The two problems I have identified in the literature regarding this aquatic application of
magnetoreception in nanoscales is first the creation of unique fields that travel through
liquids, such as the bloodstream, that contain so much interference from other atoms,
ect. The other problem is recovery of the nanobot. If the robot does the job, how will
the robot be destroyed? My previous paper suggested using ultrasound to break up the
carbon nanofibers into tiny pieces that can be excreted. So far, carbon nanotubes have
been the suggested material for the main body construction of nanobots as they do no
elicit an immune response in vertebrate hosts (SO FAR!!).
2. Thermoreception:
• Bioinspiration: I was inspired by the Fire-Seeking beetle article in this week’s Module.
The beetles have small fluid filled sensor “pits” that expand when exposed to infrared
radation. (Schmitz & Bousack. 2012). I remember from previous experience that cancer
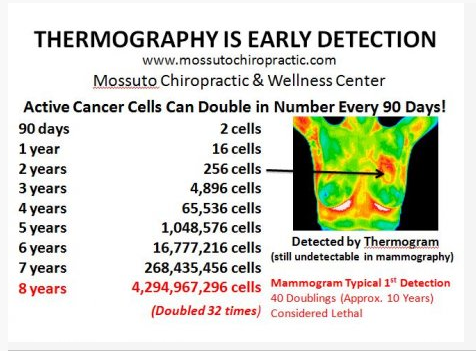
cells are generally “hotter” than normal human tissue. This concept has been used for years
in the field of thermography detection of breast cancers. The cancer tissue divides faster
than normal tissue; cancer cells also have increased amounts of insulin receptors and
metabolize glucose faster. This extra heat is used to create thermo-images of cancer: (See
figure below):
• The magnetic crystals will have two functions:
• 1. The crystals can be used to guide the device through the human body via an
applied magnetic field OR..
• 2. The crystals will detect and be attracted to a magnetic structure already within
the body.
Regarding Option #2, research has been done on producing biosensors that respond to
magnetic targets created by antibody ligand interaction. Cancer cells possess unique
surface receptors. Magnetically labeled antibodies (ferro) have been produced that attach
to cells, groups of cells in tumors creating strong magnetic fields that can be sensed by
the nanobot. The nanobot then can recognize very specific targets for some purpose such
as medicine delivery, or attach to the cells where an external field can then activate the
robot to heat up, release some cargo, ect. (See article Smith, 2011).
• Limitations:
• The two problems I have identified in the literature regarding this aquatic application of
magnetoreception in nanoscales is first the creation of unique fields that travel through
liquids, such as the bloodstream, that contain so much interference from other atoms,
ect. The other problem is recovery of the nanobot. If the robot does the job, how will
the robot be destroyed? My previous paper suggested using ultrasound to break up the
carbon nanofibers into tiny pieces that can be excreted. So far, carbon nanotubes have
been the suggested material for the main body construction of nanobots as they do no
elicit an immune response in vertebrate hosts (SO FAR!!).
2. Thermoreception:
• Bioinspiration: I was inspired by the Fire-Seeking beetle article in this week’s Module.
The beetles have small fluid filled sensor “pits” that expand when exposed to infrared
radation. (Schmitz & Bousack. 2012). I remember from previous experience that cancer
cells are generally “hotter” than normal human tissue. This concept has been used for years
in the field of thermography detection of breast cancers. The cancer tissue divides faster
than normal tissue; cancer cells also have increased amounts of insulin receptors and
metabolize glucose faster. This extra heat is used to create thermo-images of cancer: (See
figure below):
Image: http://mossutochiropractic.com/thermal-imaging/breast-cancer---thermal-imaging.html)
• How It Works:
• A nanorobot can be equipped with either liquid filled nanoshells (composed of carbon
fibers) that expand when infrared radiation reaches a threshold intensity ABOVE that of
normal human tissue or infrared detectors composed of metal that reach a threshold
resonance can elicit an electrical current that instructs the nanobot attach/destroy/
release its contents.
• Limitations:
• The biggest problem with heat sensors working within the human body is that there are
many other, non-cancerous processes that produce excess heat that can be confused
with cancerous growth. Acute inflammation due to injury or chronic inflammation due
to auto-immune diseases are two examples of situations that may limit the use of a
nanorobot with infrared detectors
3. Chemoreception:
• Bioinspiration: The chemical receptors located on cell membranes and the signal
transduction cascades these receptors initiate are shared mechanisms in every living cell.
Chemoreception is the primary mechanism through which organisms respond to
environmental cues. I mentioned this mechanism in my last post and have found in my
research that each cancer cell contains unique surface receptors. Some common receptors
shared by all human cancers are increased folic acid and insulin-like receptors amongst
others. Nanorobots can be built with specific ligands that would attach to target cells. Like
in my former post, the ligand, once attached to the receptor, would move a series of gears
by molecular conformational changes and the gears would pull forward the capsule
containing the dose of medicine or whatever cargo may be inside of the nanobot. (See
figures below).
• Video 1: Cancer Fighting Bacteriobot Video (2014) Ligand Chemistry
• A nanorobot can be equipped with either liquid filled nanoshells (composed of carbon
fibers) that expand when infrared radiation reaches a threshold intensity ABOVE that of
normal human tissue or infrared detectors composed of metal that reach a threshold
resonance can elicit an electrical current that instructs the nanobot attach/destroy/
release its contents.
• Limitations:
• The biggest problem with heat sensors working within the human body is that there are
many other, non-cancerous processes that produce excess heat that can be confused
with cancerous growth. Acute inflammation due to injury or chronic inflammation due
to auto-immune diseases are two examples of situations that may limit the use of a
nanorobot with infrared detectors
3. Chemoreception:
• Bioinspiration: The chemical receptors located on cell membranes and the signal
transduction cascades these receptors initiate are shared mechanisms in every living cell.
Chemoreception is the primary mechanism through which organisms respond to
environmental cues. I mentioned this mechanism in my last post and have found in my
research that each cancer cell contains unique surface receptors. Some common receptors
shared by all human cancers are increased folic acid and insulin-like receptors amongst
others. Nanorobots can be built with specific ligands that would attach to target cells. Like
in my former post, the ligand, once attached to the receptor, would move a series of gears
by molecular conformational changes and the gears would pull forward the capsule
containing the dose of medicine or whatever cargo may be inside of the nanobot. (See
figures below).
• Video 1: Cancer Fighting Bacteriobot Video (2014) Ligand Chemistry
• Video 2: Building Nanobots with specific shapes using DNA Origami
Note: This is part three of a three-step series. VERY AWESOME BIOLOGY TEACHERS!! This shows a direct application of DNA to physics!!
Note: This is part three of a three-step series. VERY AWESOME BIOLOGY TEACHERS!! This shows a direct application of DNA to physics!!
- Video 3: Harvard builds cancer-killing robot using DNA that finds and destroys by releasing payloads. COOL COOL!
• How it Works:
In the second video, the materials used to build the chemoreceptors are synthetic
DNA that can respond to very specific conformational changes such as those
receptors found at the cancer cell surface. Synthetic amino acids can also be
attached to the robot that detect specific surface receptors. The Self-assembly of
DNA and Amino acids in solution is capitalized on here because this is the same
process that occurs in living cells. DNA origami is a powerful method for
manufacturing precise nanoscale objects by programming the self-assembly of
DNA molecules. These “receptors” can be utilized to design a nanorobot capable
of sensing biological cues and responding by shape shifting, subsequently
relayed to a desired effect. ( Ishay, Ben. 2013).
“When they come in contact with that ligand, they bind to it and unfurl,
allowing the device to open and release its contents. “It’s kind of like the
space shuttle—you can put any payload you want in it,” Douglas explains.
“What you use is going to depend on the application.”
(Harvard Magazine, 2012).
• Limitations:
The biggest problem facing the application of chemoreception within a
robot is the creating the artificial synthesis of shape-specific ligand that
match existing cell surface proteins. The production of the robot is
difficult to direct outside of a living cell; how do you connect amino
acids or DNA nucleotides in the correct sequence without the aid of
ribosomes or enzyme? (See Video 2 and 3 above for details and
limitations). How can on attach the DNA/artifical protein ligand to an
existing robot that carries a payload is an issue but the current research
suggests making the entire robot out of artificially produced DNA popup models. Since human machines are too big to construct artificial molecules, relying on synthesis via microbes or viruses is on the table or
the creation of artificial ribosomes.
Pictures of my Nanobot with sensors:
In the second video, the materials used to build the chemoreceptors are synthetic
DNA that can respond to very specific conformational changes such as those
receptors found at the cancer cell surface. Synthetic amino acids can also be
attached to the robot that detect specific surface receptors. The Self-assembly of
DNA and Amino acids in solution is capitalized on here because this is the same
process that occurs in living cells. DNA origami is a powerful method for
manufacturing precise nanoscale objects by programming the self-assembly of
DNA molecules. These “receptors” can be utilized to design a nanorobot capable
of sensing biological cues and responding by shape shifting, subsequently
relayed to a desired effect. ( Ishay, Ben. 2013).
“When they come in contact with that ligand, they bind to it and unfurl,
allowing the device to open and release its contents. “It’s kind of like the
space shuttle—you can put any payload you want in it,” Douglas explains.
“What you use is going to depend on the application.”
(Harvard Magazine, 2012).
• Limitations:
The biggest problem facing the application of chemoreception within a
robot is the creating the artificial synthesis of shape-specific ligand that
match existing cell surface proteins. The production of the robot is
difficult to direct outside of a living cell; how do you connect amino
acids or DNA nucleotides in the correct sequence without the aid of
ribosomes or enzyme? (See Video 2 and 3 above for details and
limitations). How can on attach the DNA/artifical protein ligand to an
existing robot that carries a payload is an issue but the current research
suggests making the entire robot out of artificially produced DNA popup models. Since human machines are too big to construct artificial molecules, relying on synthesis via microbes or viruses is on the table or
the creation of artificial ribosomes.
Pictures of my Nanobot with sensors:
SOURCES:
Ashammakhi, N. "Nanosize, Mega-Impact, Potential for Medical Applications of
Nanotechnology." Journal of Craniofacial Surgery 17.1 (2006): 3-7.
Eder, S. H. K., H. Cadiou, A. Muhamad, P. A. Mcnaughton, J. L. Kirschvink, and M. Winklhofer.
"Magnetic Characterization of Isolated Candidate Vertebrate Magnetoreceptor Cells."
Proceedings of the National Academy of Sciences 109.30 (2012): 2022-2027.
Greenfield D; McEvoy AL; Shroff H; Crooks GE; Wingreen NS; Betzig E; Liphardt J. 2009.
Self-organization of the Escherichia coli chemotaxis network imaged with super-resolution light
microscopy. PLoS Biology (from www.AskNature.org)
H. Tung, D. F. Sargent, B. J. Nelson, "Protein crystal harvesting using the RodBot: a wireless
mobile microrobot", Journal of Applied Crystallography, Vol. 4, 2014, pp. 692-70
Harvard Magazine: “Nanorobots fight Cancer” http://harvardmagazine.com/2012/09/cancerfighting-
robots
Ben-Ishay, Eldad, Almogit Abu-Horowitz, and Ido Bachelet. "Designing a Bio-responsive Robot
from DNA Origami." Journal of Visualized Experiments 77 (2013)
KUTERBACH, D. A. AND WALCOTT, B. (1986a). Iron-containing cells in the honey-bee
( Apis mellifera ).I. Adult morphology and physiology. J. exp. Biol. 126, 375–387.
KUTERBACH, D. A. AND WALCOTT, B. (1986b). Iron-containing cells in the honey-bee
(Apis mellifera).
II. Accumulation during development. J. exp. Biol. 126, 389–401.
Peer, Dan, Jeffrey M. Karp, Seungpyo Hong, Omid C. Farokhzad, Rimona Margalit, and Robert
Langer. "Nanocarriers as an Emerging Platform for Cancer Therapy." Nature Nanotechnology
2.12 (2007): 751-60.
Mavroidis, Constantinos, Antoine Ferreira, and Constantinos Mavroidis. "Chapter 14: Generating
Magnetic Fields for Controlling Nanorobots in Medical Applications." Nanorobotics: Current
Approaches and Techniques. N.p.: Springer Science Media, 2013. 275-97.
"Nanotechnology in Cancer Treatment." Nanotechnology in Cancer Treatment. Hawks-Perch
Technical Writing, 2014. Web. 8 July 2014. <http://www.understandingnano.com/cancertreatment-
nanotechnology.html>.
"Overview of the Multi-Scale Robotics Lab." Institute of Robotics and Intelligent Systems. Swiss
Federal Institue of Technology, ETH, 2014. Web. 8 July 2014. <http://www.iris.ethz.ch/msrl/>.
Schuerle, B. E. Kratochvil, S. Pané, A. M. Zeeshan, B. J. Nelson, "Generating Magnetic Fields
for Controlling Nanorobots in Medical Applications", Nanorobotics, Springer Science+Business
Media, Berlin, Heidelberg, New York, 2013, pp 275-299.
DAWN SASEK Module 4: Add Sensors to your Robot
Smith, Joshua E., Kim E. Sapsford, Weihong Tan, and Frances S. Ligler. "Optimization of
Antibody-conjugated Magnetic Nanoparticles for Target Preconcentration and Immunoassays."
Analytical Biochemistry 410.1 (2011): 124-32.
Schmitz & Bousack. 2012. Modelling a Historic Oil-Tank Fire Allows an Estimation of the
Sensitivity of the Infrared Receptors in Pyrophilous Melanophila Beetles. PLoS ONE http://
dx.doi.org/10.1371/journal.pone.0037627
"Thermography in Breast Cancer Testing." Thermography in Breast Cancer Testing. N.p., n.d.
Web. 8 July 2014. <http://www.a-zbreastcancer.com/articles/thermography.htm>.
Ashammakhi, N. "Nanosize, Mega-Impact, Potential for Medical Applications of
Nanotechnology." Journal of Craniofacial Surgery 17.1 (2006): 3-7.
Eder, S. H. K., H. Cadiou, A. Muhamad, P. A. Mcnaughton, J. L. Kirschvink, and M. Winklhofer.
"Magnetic Characterization of Isolated Candidate Vertebrate Magnetoreceptor Cells."
Proceedings of the National Academy of Sciences 109.30 (2012): 2022-2027.
Greenfield D; McEvoy AL; Shroff H; Crooks GE; Wingreen NS; Betzig E; Liphardt J. 2009.
Self-organization of the Escherichia coli chemotaxis network imaged with super-resolution light
microscopy. PLoS Biology (from www.AskNature.org)
H. Tung, D. F. Sargent, B. J. Nelson, "Protein crystal harvesting using the RodBot: a wireless
mobile microrobot", Journal of Applied Crystallography, Vol. 4, 2014, pp. 692-70
Harvard Magazine: “Nanorobots fight Cancer” http://harvardmagazine.com/2012/09/cancerfighting-
robots
Ben-Ishay, Eldad, Almogit Abu-Horowitz, and Ido Bachelet. "Designing a Bio-responsive Robot
from DNA Origami." Journal of Visualized Experiments 77 (2013)
KUTERBACH, D. A. AND WALCOTT, B. (1986a). Iron-containing cells in the honey-bee
( Apis mellifera ).I. Adult morphology and physiology. J. exp. Biol. 126, 375–387.
KUTERBACH, D. A. AND WALCOTT, B. (1986b). Iron-containing cells in the honey-bee
(Apis mellifera).
II. Accumulation during development. J. exp. Biol. 126, 389–401.
Peer, Dan, Jeffrey M. Karp, Seungpyo Hong, Omid C. Farokhzad, Rimona Margalit, and Robert
Langer. "Nanocarriers as an Emerging Platform for Cancer Therapy." Nature Nanotechnology
2.12 (2007): 751-60.
Mavroidis, Constantinos, Antoine Ferreira, and Constantinos Mavroidis. "Chapter 14: Generating
Magnetic Fields for Controlling Nanorobots in Medical Applications." Nanorobotics: Current
Approaches and Techniques. N.p.: Springer Science Media, 2013. 275-97.
"Nanotechnology in Cancer Treatment." Nanotechnology in Cancer Treatment. Hawks-Perch
Technical Writing, 2014. Web. 8 July 2014. <http://www.understandingnano.com/cancertreatment-
nanotechnology.html>.
"Overview of the Multi-Scale Robotics Lab." Institute of Robotics and Intelligent Systems. Swiss
Federal Institue of Technology, ETH, 2014. Web. 8 July 2014. <http://www.iris.ethz.ch/msrl/>.
Schuerle, B. E. Kratochvil, S. Pané, A. M. Zeeshan, B. J. Nelson, "Generating Magnetic Fields
for Controlling Nanorobots in Medical Applications", Nanorobotics, Springer Science+Business
Media, Berlin, Heidelberg, New York, 2013, pp 275-299.
DAWN SASEK Module 4: Add Sensors to your Robot
Smith, Joshua E., Kim E. Sapsford, Weihong Tan, and Frances S. Ligler. "Optimization of
Antibody-conjugated Magnetic Nanoparticles for Target Preconcentration and Immunoassays."
Analytical Biochemistry 410.1 (2011): 124-32.
Schmitz & Bousack. 2012. Modelling a Historic Oil-Tank Fire Allows an Estimation of the
Sensitivity of the Infrared Receptors in Pyrophilous Melanophila Beetles. PLoS ONE http://
dx.doi.org/10.1371/journal.pone.0037627
"Thermography in Breast Cancer Testing." Thermography in Breast Cancer Testing. N.p., n.d.
Web. 8 July 2014. <http://www.a-zbreastcancer.com/articles/thermography.htm>.



 RSS Feed
RSS Feed