The following is an assignment where the idea of harnessing the sun via synthetic photosynthesis mechanisms is explored. One of the coolest resources so far encountered in this course was utilized in this lesson. The issue of ZYGOTE QUARTERLY are particularly awesome and useful for both scientists and citizens. See the link below for a direct route to the magazine website.
Module 5 Assignment: Artificial Photosynthesis
Prologue:
Before I begin my official introduction to Photosynthesis, I would like to address some issues that I have encountered in my teaching of photosynthesis over the past two decades. First, I think that all of us teachers in IB 535 approach photosynthesis very differently depending on which level of biology/science we teach. AP Biology covers the details of the z-scheme of the light reactions including all major enzymes and molecules important in electron transport. Other classes such as introductory biology or plant science simply cover the basics with emphasis on the overall nutrient and energy cycling involved in the process. With the new NGSS standards upon us, where students are being asked to understand the CONCEPTS and IMPACT behind such important processes as photosynthesis on their everyday lives, I think this artificial photosynthesis concept is a great way to stimulate student interest while at the same time teaching them “the big picture” and importance of the process. Second, I would like to express how happy I am that scientists are using more physical means of created light-dependent fuels instead of using genetically modified microorganisms. There was much research done on genetically modifying photosynthetic bacteria to produce the hydrogen/biofuels in “green tank reactors”; as a biologist, I worried about the release of these “exotic” organisms into the wild, and the effects on the natural balanced ecosystems. I am excited to explore the application of the physical methods in the production of biofuels and think this could be a great topic to be used in the classroom. (Why are physical methods better that the genetic modification methods?).
(Side Note: The main genetic modifications being done in the previous decade included the production of photosynthetic bacteria whose pigments had shorter “membrane insertion stalks” so that light could penetrate deeper within an aquatic bioreactor. These nanoscale differences in chlorophyll structure can be used to create length-tailored, functional pigments to maximize light penetration in bioreactors that use aquatic, photosynthetic microbes. HERE IS LINK TO ARTICLE RELATED TO THIS CONCEPT)
Introduction:
Besides the nuclear fusion reaction that occurs within stars, the chemistry and physics associated with the process of photosynthesis is undeniably the most important concept associated with the survival of all life on planet Earth. Every human society of ancient times celebrated and embraced the arrival of the sun. Ancient people’s lives were set in “real carbon-time”, that is, were dependent of the production of materials from plants on a seasonal basis and had yet to tap into “stored-carbon” of fossil fuels. Because of this, there was real connection made between the presence of the sun and growth of crops, clothes, animals, ect. (Link to fun interviews from Veritasium videos on misconceptions of photosynthesis)
Prologue:
Before I begin my official introduction to Photosynthesis, I would like to address some issues that I have encountered in my teaching of photosynthesis over the past two decades. First, I think that all of us teachers in IB 535 approach photosynthesis very differently depending on which level of biology/science we teach. AP Biology covers the details of the z-scheme of the light reactions including all major enzymes and molecules important in electron transport. Other classes such as introductory biology or plant science simply cover the basics with emphasis on the overall nutrient and energy cycling involved in the process. With the new NGSS standards upon us, where students are being asked to understand the CONCEPTS and IMPACT behind such important processes as photosynthesis on their everyday lives, I think this artificial photosynthesis concept is a great way to stimulate student interest while at the same time teaching them “the big picture” and importance of the process. Second, I would like to express how happy I am that scientists are using more physical means of created light-dependent fuels instead of using genetically modified microorganisms. There was much research done on genetically modifying photosynthetic bacteria to produce the hydrogen/biofuels in “green tank reactors”; as a biologist, I worried about the release of these “exotic” organisms into the wild, and the effects on the natural balanced ecosystems. I am excited to explore the application of the physical methods in the production of biofuels and think this could be a great topic to be used in the classroom. (Why are physical methods better that the genetic modification methods?).
(Side Note: The main genetic modifications being done in the previous decade included the production of photosynthetic bacteria whose pigments had shorter “membrane insertion stalks” so that light could penetrate deeper within an aquatic bioreactor. These nanoscale differences in chlorophyll structure can be used to create length-tailored, functional pigments to maximize light penetration in bioreactors that use aquatic, photosynthetic microbes. HERE IS LINK TO ARTICLE RELATED TO THIS CONCEPT)
Introduction:
Besides the nuclear fusion reaction that occurs within stars, the chemistry and physics associated with the process of photosynthesis is undeniably the most important concept associated with the survival of all life on planet Earth. Every human society of ancient times celebrated and embraced the arrival of the sun. Ancient people’s lives were set in “real carbon-time”, that is, were dependent of the production of materials from plants on a seasonal basis and had yet to tap into “stored-carbon” of fossil fuels. Because of this, there was real connection made between the presence of the sun and growth of crops, clothes, animals, ect. (Link to fun interviews from Veritasium videos on misconceptions of photosynthesis)
Today, much of that connection has been lost to the general public as we live our lives in artificial environments, using the stored energy in the bonds of fossil fuels to produce quick, seasonally-independent sources of electricity, clothes, foods, medicines, containers, building materials and the list goes on. It is not surprising that modern-day teenagers and adults struggle to connect the process of photosynthesis with their “high-tech” synthetic clothing, the plastic bottles they drink from, the medicines they consume, ect. (There is a great video called “The 11th Hour, narrated by Leonardo DiCaprio, that includes interviews with famous scientists. Great to show some clips from this in the classroom; the movie does a good job addressing the “real-time” carbon issue. LINK HERE)
I believe it is important that we make photosynthesis very “real” to them by pointing out how important ancient sunlight is to every aspect of their modern lives, then stress the finite nature of the supply of this energy source.
I believe it is important that we make photosynthesis very “real” to them by pointing out how important ancient sunlight is to every aspect of their modern lives, then stress the finite nature of the supply of this energy source.
Inspiration:
I think all of the choices offered in Module 5 concerning artificial photosynthesis sound exciting. As mentioned in the prologue, I have chosen to focus on the use of artificial catalysts in non-living, physical systems as opposed to the applications in modified living systems out of concern for possible adverse affects on the ecosystem if any of the modified organisms were accidentally released. I have chosen to focus on the work done by Meyer on the use of artificial catalysts, mounted on silicates or some other inexpensive surface, to catalyze both the generation of hydrogen from splitting water, and the production of methanol from carbon dioxide. As mentioned in one paper by Meyer,
“In order for solar energy to serve as a primary energy source, it must be paired with energy storage on a massive scale.” (Meyer, 2011).
In short, the goal of the artificial catalyst research can be summed by this excerpt from a research paper published by one of Meyers’ groups in 2005:
"The molecular complexity of the natural photosynthetic apparatus is both an object lesson that illustrates the difficulties involved and an inspiration to research in this area. With successful application, artificial photosynthesis would shortcut the billions of years of evolution that led to working photosynthetic membranes.”
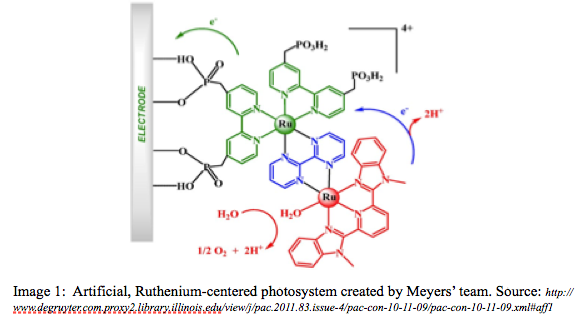
Solar panels work mainly when the sun is shining. Storing the energy produced from this process requires very expensive batteries that take up much space. Since the sun doesn’t shine constantly, this storage dilemma is of critical concern. Although solar cells are much more efficient at converting solar into electric energy, the PROBLEM of storing this energy for future use has only been worked out by natural photosynthesis in the form of organic compounds. (National Research Council, 2010). Dr. Meyer and his research group have created catalysts that have reactive centers similar to those found in the photosystems of plants. See Images 1 and 2 below):
- photosystems of plants, bacteria, archae synthetic photosynthetic catalysts
- bypass billions of years of evolution to simplify steps in photosynthsis
- recreating nanoscale, atomic level electron transfer is extremely challenging!!
- coupling the oxidative and reduction events
- relies on high levels of dissolved, aqueous CO2 passing across membrane; where do find these conditions naturally???
I think all of the choices offered in Module 5 concerning artificial photosynthesis sound exciting. As mentioned in the prologue, I have chosen to focus on the use of artificial catalysts in non-living, physical systems as opposed to the applications in modified living systems out of concern for possible adverse affects on the ecosystem if any of the modified organisms were accidentally released. I have chosen to focus on the work done by Meyer on the use of artificial catalysts, mounted on silicates or some other inexpensive surface, to catalyze both the generation of hydrogen from splitting water, and the production of methanol from carbon dioxide. As mentioned in one paper by Meyer,
“In order for solar energy to serve as a primary energy source, it must be paired with energy storage on a massive scale.” (Meyer, 2011).
In short, the goal of the artificial catalyst research can be summed by this excerpt from a research paper published by one of Meyers’ groups in 2005:
"The molecular complexity of the natural photosynthetic apparatus is both an object lesson that illustrates the difficulties involved and an inspiration to research in this area. With successful application, artificial photosynthesis would shortcut the billions of years of evolution that led to working photosynthetic membranes.”
Solar panels work mainly when the sun is shining. Storing the energy produced from this process requires very expensive batteries that take up much space. Since the sun doesn’t shine constantly, this storage dilemma is of critical concern. Although solar cells are much more efficient at converting solar into electric energy, the PROBLEM of storing this energy for future use has only been worked out by natural photosynthesis in the form of organic compounds. (National Research Council, 2010). Dr. Meyer and his research group have created catalysts that have reactive centers similar to those found in the photosystems of plants. See Images 1 and 2 below):
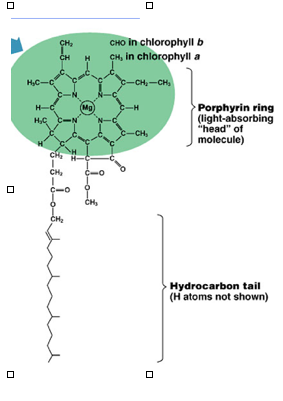
Image 2: Structure of Chlorophyll b from natural
world. Note the similar ligand-central metal
ion structure. Image Source:
http://agron-www.agron.iastate.edu/Courses/Agron317/Pigment_Inhibitors.htm
world. Note the similar ligand-central metal
ion structure. Image Source:
http://agron-www.agron.iastate.edu/Courses/Agron317/Pigment_Inhibitors.htm
What I found so intriguing about Meyer’s work was that the catalyst his team developed did two steps in one, that is, the same Ruthenium-centered catalyst was able to both split water to produce hydrogen ions (as occurs in natural photosynthesis LIGHT REACTIONS) AND split carbon dioxide into carbon monoxide and oxygen gas. The carbon monoxide can be used to produce methanol by somehow adding the hydrogen released in the first step to the carbon monoxide. (DARK REACTIONS or CALVIN CYCLE) This is the problem..how to do this?? Although the group was able to accomplish the baseline chemistry associated with the major steps of photosynthesis, the part yet to be one is combining the two steps to produce the final product: a reduced organic compound, like a sugar, or methanol, that can store the energy of the light capture for future use. This would be like the the NADP+/NADPH and enzyme cascades coupling the light and dark reactions during natural photosynthesis.
Teaching Methods:
Since my post is getting pretty wordy here, I am going to focus on how I would introduce photosynthesis as a critical process, then how I would relate photosynthesis to a “physical machine” using concepts associated with electricity, chemistry and biology.
Day 1: BRAIN-STORMING
Activity:
Day 2-5: DEMONSTRATIONS and MINI-Experiments
Demonstration List:
Demonstration #1: The photoelectric effect. If your physics teacher does not have an apparatus to demonstrate this, here are a few videos you can use. One is from PHET and another is from a youtube video. The reason I show this to them is to lead them up to understanding that photosynthesis is really a physical phenomenon that relies on modifications of the photoelectric effect: Light shines on a metal and electrons are excited. This will help them understand why there is always some metal at the center of the artificial and natural photosynthetic systems.
PHET Photoelectric effect Simulation
This simulation is very fun; you can play around with it and use as a quick animated guide to what the photoelectric effect is...creating a current using light and a metal.
Teaching Methods:
Since my post is getting pretty wordy here, I am going to focus on how I would introduce photosynthesis as a critical process, then how I would relate photosynthesis to a “physical machine” using concepts associated with electricity, chemistry and biology.
Day 1: BRAIN-STORMING
Activity:
- lay out examples of plastic products students use everyday (bottles, packaging, cosmetic cases, ect), empty aspirin bottles (to identify that medicines use fossil fuel products for feedstock), an out of season food item (to demonstrate that fossil fuels help bring these items to them off-season...like a tomato or grape in January), examples of foods derived directly by plants (chocolate, coffee, ect) and meat-based foods. (OF COURSE MAY USE PHOTOS TO SAVE MONEY), any heavy, cast-iron object like a faucet, or baking dish (the power to melt the metal and form in to the shapes requires tons of energy derived from fossil fuels). (Or any idea you can add to the list)
- Place students in groups. Ask each team to explain how each item, ultimately, owes its production to the power of the sun.
Have students share their ideas with the class. Look for patterns, misconceptions about the relationships. This is a great opportunity to get a feel for how much your students know about the process; lay down some basic foundations as to how every aspect of their relates to the energy of the sun.
Check off each student’ sheet for participation points.
Day 2-5: DEMONSTRATIONS and MINI-Experiments
Demonstration List:
Demonstration #1: The photoelectric effect. If your physics teacher does not have an apparatus to demonstrate this, here are a few videos you can use. One is from PHET and another is from a youtube video. The reason I show this to them is to lead them up to understanding that photosynthesis is really a physical phenomenon that relies on modifications of the photoelectric effect: Light shines on a metal and electrons are excited. This will help them understand why there is always some metal at the center of the artificial and natural photosynthetic systems.
PHET Photoelectric effect Simulation
This simulation is very fun; you can play around with it and use as a quick animated guide to what the photoelectric effect is...creating a current using light and a metal.
- Demonstration/Experiment #2: Students can build their own battery powered electric currents that can be used to split water into hydrogen and oxygen gas. Here is a video clip of a very simple experiment you can set up with your students to show how electricity is used to split water:
- Building mini-water splitter
- You can have students collect gas over the electrodes and do the flame-test to see positive test for hydrogen gas production. One is electrode produces oxygen, the other, hydrogen...just like plants.
Demonstration/Experiment #3:
- Have students do the experiment with aquatic plants and oxygen production. There are several ways to do this..I do two experiments with my students (AP/Bio/regular bio). Bozeman biology video has great video clip explaining the experiment:
- Photosynthesis Experiment: Wavelength of light and oxygen production.
- Here you can change up wavelengths by placing different colored bulbs in light source. I use blue, red, black lights, yellow, green and white. Very cool..relate the oxygen bubble production to the same battery experiment done the day before. Explain the similarities..how are plants more efficient at producing oxygen and hydrogen than the battery method? Where does all of the oxygen you breath come from? Relate this to the photosynthesis experiment.
- Why is photosynthesis so important for our oxygen needs?
Comparison Video/Discussion: This is a video clip from Meyer’s Ruthenium-based catalyst in action. I would like to show this to my students after they have done the battery-generated splitting of water AND the plant-generated splitting of water and have them compare these two with what they see in this video. I am hoping that they notice that water is being split WITHOUT an external electrical source.
This is as far as I got on this...I am stopping here because I am blabbing too much..I need to somehow pair the splitting of water with the carbon aspect...I have included a brief possibility of how I might address the carbon issue below. LINK TO VIDEO CLIP
This is as far as I got on this...I am stopping here because I am blabbing too much..I need to somehow pair the splitting of water with the carbon aspect...I have included a brief possibility of how I might address the carbon issue below. LINK TO VIDEO CLIP
Demonstration #4: Burning experiments. So far the kids get the idea that oxygen comes from photosynthesis via “light hydrolysis” of water...what about the carbon? I usually burn several items in front of class in my fume hood....sugars (AND ALL FOOD ITEMS THAT ARE DRIED.. textiles, plastic bottles, ect...ALL ITEMS TURN BLACK...what element is this???? Most students realize that this is carbon from previous experience. The question is ..where did the carbon come from? How do plants get the carbon connected to the hydrogen... THIS IS THE CHALLENGE!!
Summary:
To summarize, I prefer the non-biological approach, not because I am against the use of organisms for energy production, it is the genetic modifications that I find somewhat scary from an ecological standpoint. I an enjoying the robotic applications of the biological concepts; I think this is a safer way of employing biological processes without the challenges of managing a self-replicating “entity” that may escape from cultivation.
I hope you enjoyed the post and I apologize for the size of this file!! :)
PS: Here is a link to an amazing page with graphics that you can use to show your class application of solar fuel technology: http://www.rsc.org/ScienceAndTechnology/Policy/Documents/solar-fuels-production-infographics.asp
Sources:
Song, Wenjing, Zuofeng Chen, M. Kyle Brennaman, Javier J. Concepcion, Antonio Otávio T. Patrocinio, Neyde Y. Murakami Iha, and Thomas J. Meyer. "Making Solar Fuels by Artificial Photosynthesis." Pure and Applied Chemistry 83.4 (2011): 749-68.
National Research Council of the National Academies. Hidden Costs of Energy: Unpriced Consequences of Energy Production and Use (2010).
Gust, Devens, Thomas A. Moore, and Ana L. Moore. "Solar Fuels via Artificial Photosynthesis." Accounts of Chemical Research 42.12 (2009): 1890-898.
"Solar Fuels and Artificial Photosynthesis Infographics." Solar Fuels and Artificial Photosynthesis Infographics. Royal Society of Chemistry, n.d. Web. 15 July 2014
Alstrum-Acevedo, James H., M. Kyle Brennaman, and Thomas J. Meyer. "Chemical Approaches to Artificial Photosynthesis. 2." Inorganic Chemistry 44.20 (2005): 6802-827.
VIDEOS:
The photoelectric effect: http://www.youtube.com/watch?v=0qKrOF-gJZ4
Video of catalyst splitting water WITH battery:
http://www.youtube.com/watch?v=HQ9Fhd7P_HA
Video of the catalyst splitting water WITHOUT being connected to a battery
http://www.nature.com/nature/newsvideo/suncat.mov
Summary:
To summarize, I prefer the non-biological approach, not because I am against the use of organisms for energy production, it is the genetic modifications that I find somewhat scary from an ecological standpoint. I an enjoying the robotic applications of the biological concepts; I think this is a safer way of employing biological processes without the challenges of managing a self-replicating “entity” that may escape from cultivation.
I hope you enjoyed the post and I apologize for the size of this file!! :)
PS: Here is a link to an amazing page with graphics that you can use to show your class application of solar fuel technology: http://www.rsc.org/ScienceAndTechnology/Policy/Documents/solar-fuels-production-infographics.asp
Sources:
Song, Wenjing, Zuofeng Chen, M. Kyle Brennaman, Javier J. Concepcion, Antonio Otávio T. Patrocinio, Neyde Y. Murakami Iha, and Thomas J. Meyer. "Making Solar Fuels by Artificial Photosynthesis." Pure and Applied Chemistry 83.4 (2011): 749-68.
National Research Council of the National Academies. Hidden Costs of Energy: Unpriced Consequences of Energy Production and Use (2010).
Gust, Devens, Thomas A. Moore, and Ana L. Moore. "Solar Fuels via Artificial Photosynthesis." Accounts of Chemical Research 42.12 (2009): 1890-898.
"Solar Fuels and Artificial Photosynthesis Infographics." Solar Fuels and Artificial Photosynthesis Infographics. Royal Society of Chemistry, n.d. Web. 15 July 2014
Alstrum-Acevedo, James H., M. Kyle Brennaman, and Thomas J. Meyer. "Chemical Approaches to Artificial Photosynthesis. 2." Inorganic Chemistry 44.20 (2005): 6802-827.
VIDEOS:
The photoelectric effect: http://www.youtube.com/watch?v=0qKrOF-gJZ4
Video of catalyst splitting water WITH battery:
http://www.youtube.com/watch?v=HQ9Fhd7P_HA
Video of the catalyst splitting water WITHOUT being connected to a battery
http://www.nature.com/nature/newsvideo/suncat.mov


 RSS Feed
RSS Feed